|
5/30/2023 0 Comments Equilibrium chemistry calculator 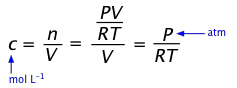
(a) \begin_3(g)Īn equilibrium mixture of NH 3( g), H 2( g), and N 2( g) at 500 ☌ was found to contain 1.35 M H 2, 1.15 M N 2, and 4. On heating, ammonia reversibly decomposes into nitrogen and hydrogen according to this equation:ĭetermining Relative Changes in ConcentrationĬomplete the changes in concentrations for each of the following reactions. We use the decomposition of ammonia as an example. Calculate the value of the Equilibrium Constant for this reaction at 200C. In this section we will see that we can relate these changes to each other using the coefficients in the balanced chemical equation describing the system. or something to that effect, it is a Type 1 Calculation. This algebraic approach to equilibrium calculations will be explored in this section.Ĭhanges in concentrations or pressures of reactants and products occur as a reaction system approaches equilibrium. Chemical is the condition of a chemical process in which both the reactants and products are present in concentrations that have no further tendency to vary with time, resulting in no apparent change in the system’s characteristics. We do so by evaluating the ways that the concentrations of products and reactants change as a reaction approaches equilibrium, keeping in mind the stoichiometric ratios of the reaction. 
While we have learned to identify in which direction a reaction will shift to reach equilibrium, we want to extend that understanding to quantitative calculations. Thus, we can use the mathematical expression for Q to determine a number of quantities associated with a reaction at equilibrium or approaching equilibrium. We know that at equilibrium, the value of the reaction quotient of any reaction is equal to its equilibrium constant. Solve for the change and the equilibrium concentrations. The balanced equation for the decomposition of PCl 5 is Because only the reactant is present initially, Qc 0, and the reaction will proceed to the right. Use algebra to perform various types of equilibrium calculations Determine the direction the reaction proceeds.Write equations representing changes in concentration and pressure for chemical species in equilibrium systems.Visit the go-to place for formulae of various concepts on and learn the concepts effortlessly. Decrease of pressure (from less moles to more moles)Ĩ.Increase of pressure (from more moles to less moles).If in any heterogenous equilibrium solid substance is also present then its active mass
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
